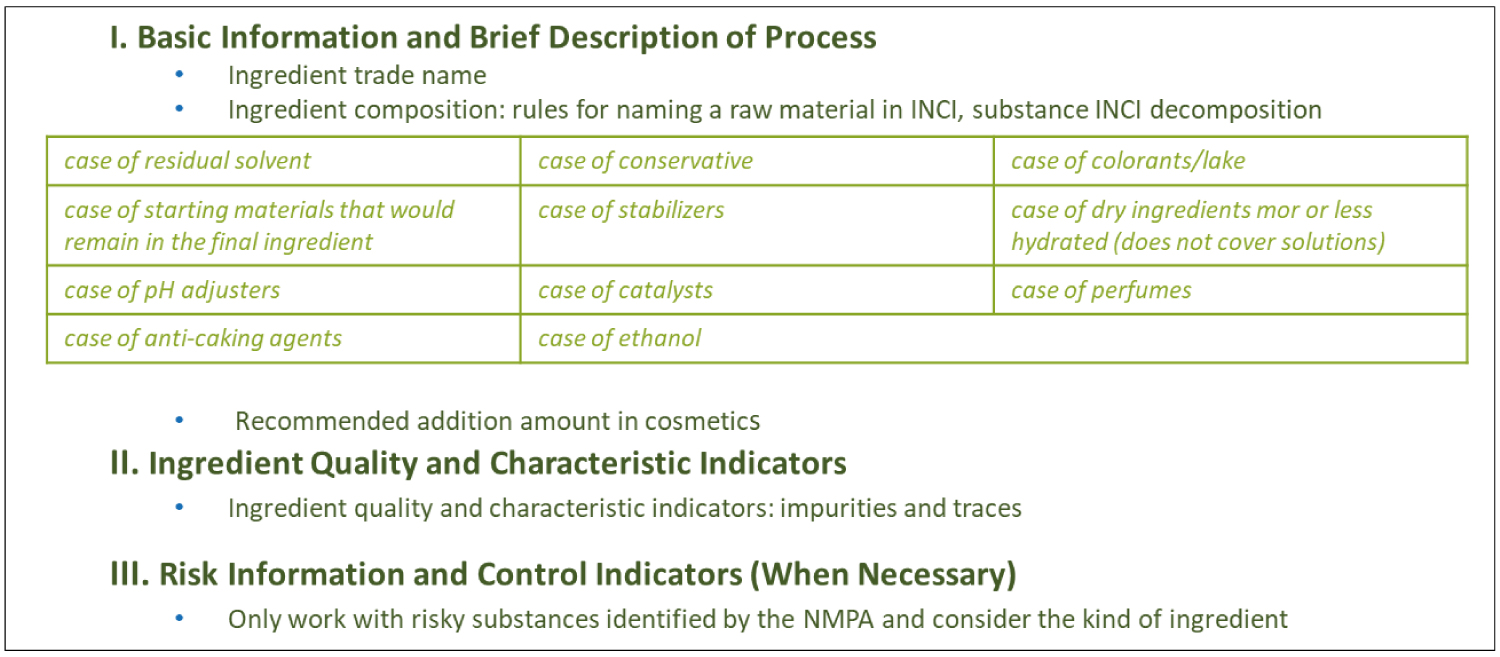
Welcome to INGRECOS Practical Guides Page
We provide you with a series of essential documents to help you navigate the regulations and best practices of the cosmetic ingredients industry. Whether you are a seasoned professional or new to the sector, these guides will give you the information you need to ensure compliance and operational excellence.
ABS Country Profiles
(Access and Benefit-Sharing)
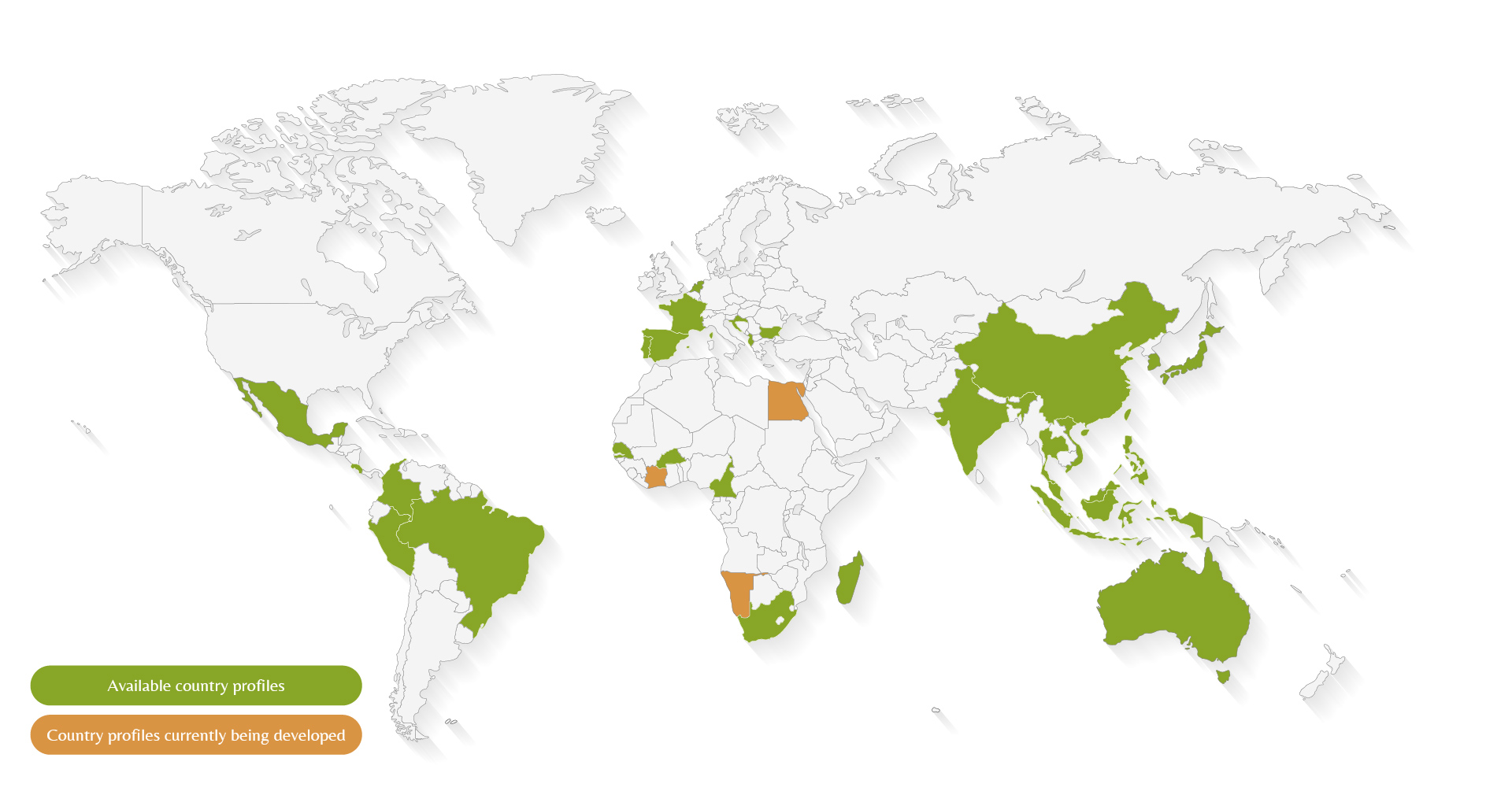
We have initiated the co-development of ABS country fact sheets with FEBEA, UNITIS, UEBT, and FIAB (with a focus on Africa) to help our members navigate the requirements of the Nagoya Protocol. These sheets are structured into 21 sections and include key questions addressed to National Competent Authorities and Focal Points.
The map below summarizes the country profiles available to our members:
EFfCI Position Papers
Our European association, EFfCI, has published numerous position papers to guide ingredient manufacturers on key issues affecting cosmetic ingredients.
You can freely access these position papers—covering topics such as Zinc Oxide Granulometric Characterization, Microplastics, GMOs, INCI vs. REACH, Titanium Dioxide (nano), and more—on the EFfCI website.
All EFfCI Position Papers are available on the INGRECOS members' extranet.
Feel free to explore these resources, and do not hesitate to contact us for any questions or further assistance!